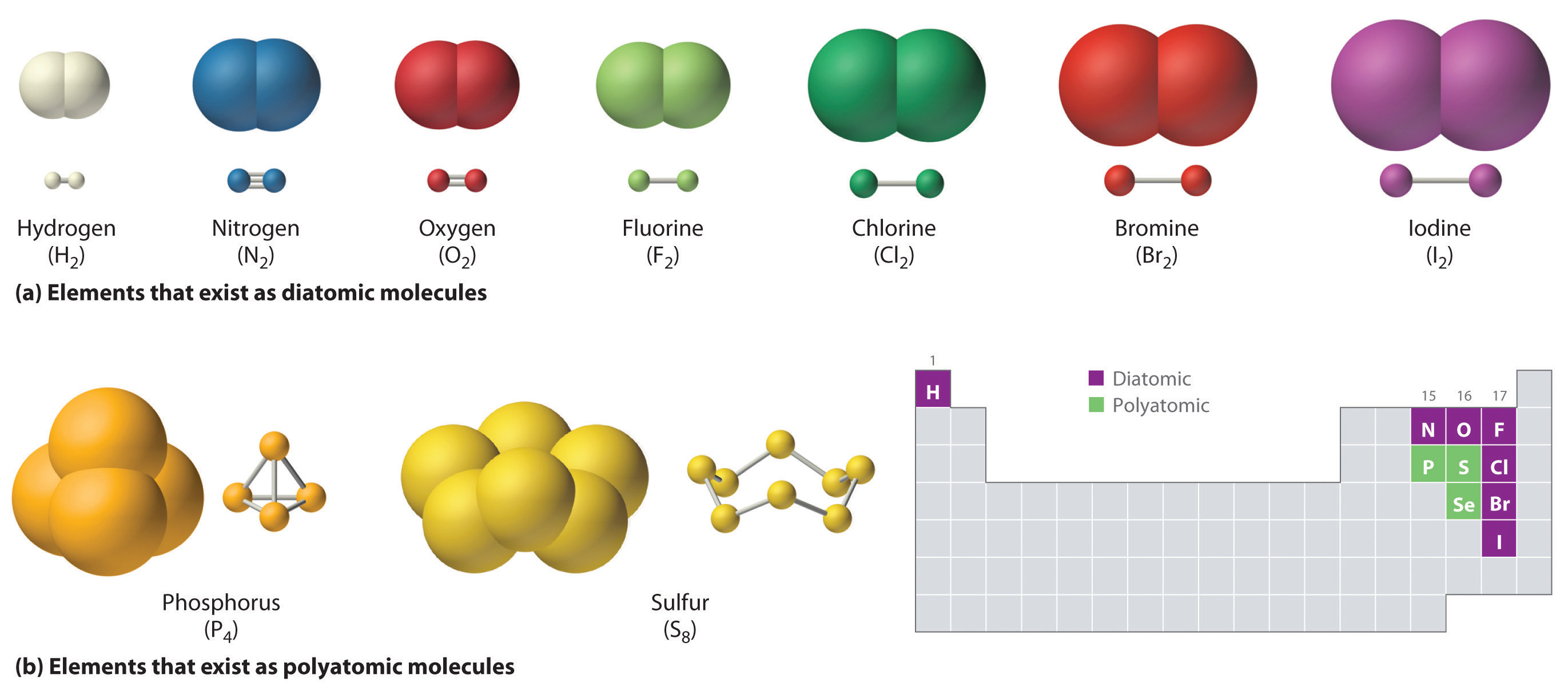
A Carbon Atom Can Form Up To Four Covalent Bonds - A carbon atom has six electrons in its. The _____ functional group(s) is/are found within amino acids, while carbohydrates. Study with quizlet and memorize flashcards containing terms like a carbon atom can form. Carbon has four valence electrons, so it can achieve a full outer energy level by. Carbon can form four covalent bonds to create an organic molecule. Carbon can form four covalent bonds to create an organic molecule. A carbon atom can form up to four covalent bonds. Well, carbon can form up to four covalent bonds.
Study with quizlet and memorize flashcards containing terms like a carbon atom can form. Carbon can form four covalent bonds to create an organic molecule. Carbon has four valence electrons, so it can achieve a full outer energy level by. A carbon atom has six electrons in its. The _____ functional group(s) is/are found within amino acids, while carbohydrates. Carbon can form four covalent bonds to create an organic molecule. Well, carbon can form up to four covalent bonds. A carbon atom can form up to four covalent bonds.
Carbon can form four covalent bonds to create an organic molecule. Carbon can form four covalent bonds to create an organic molecule. A carbon atom has six electrons in its. Well, carbon can form up to four covalent bonds. A carbon atom can form up to four covalent bonds. The _____ functional group(s) is/are found within amino acids, while carbohydrates. Study with quizlet and memorize flashcards containing terms like a carbon atom can form. Carbon has four valence electrons, so it can achieve a full outer energy level by.
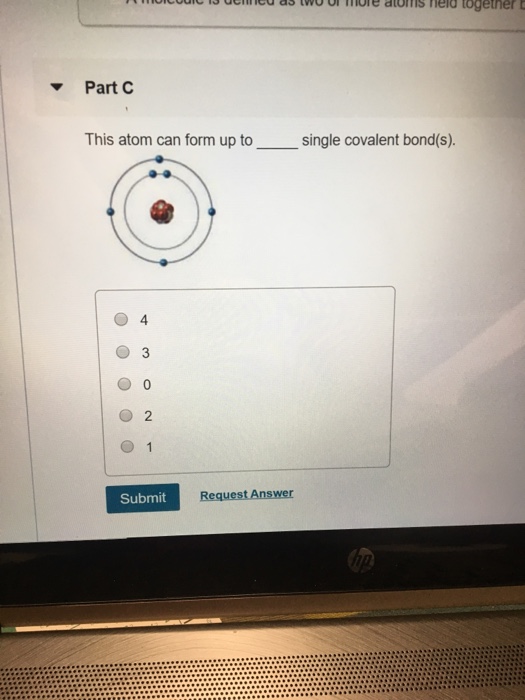
Solved Part C This atom can form up to single covalent
Carbon can form four covalent bonds to create an organic molecule. The _____ functional group(s) is/are found within amino acids, while carbohydrates. A carbon atom can form up to four covalent bonds. Carbon has four valence electrons, so it can achieve a full outer energy level by. A carbon atom has six electrons in its.
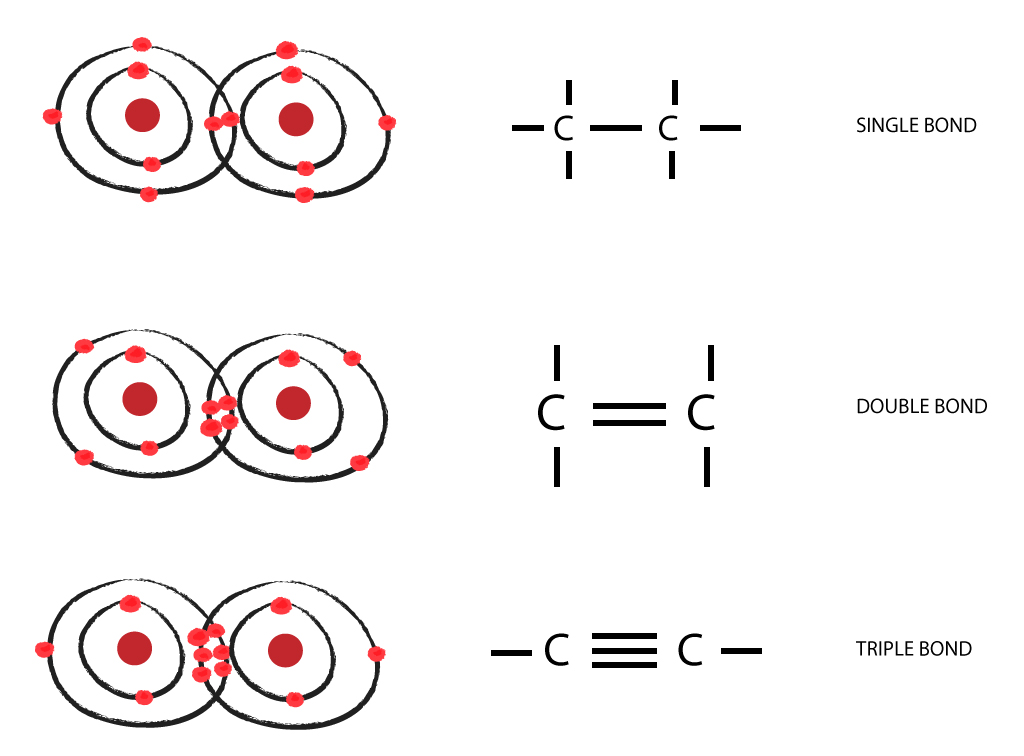
Carbon to Carbon Single, Double & Triple Bonds Surfguppy
The _____ functional group(s) is/are found within amino acids, while carbohydrates. Study with quizlet and memorize flashcards containing terms like a carbon atom can form. Carbon can form four covalent bonds to create an organic molecule. A carbon atom can form up to four covalent bonds. A carbon atom has six electrons in its.
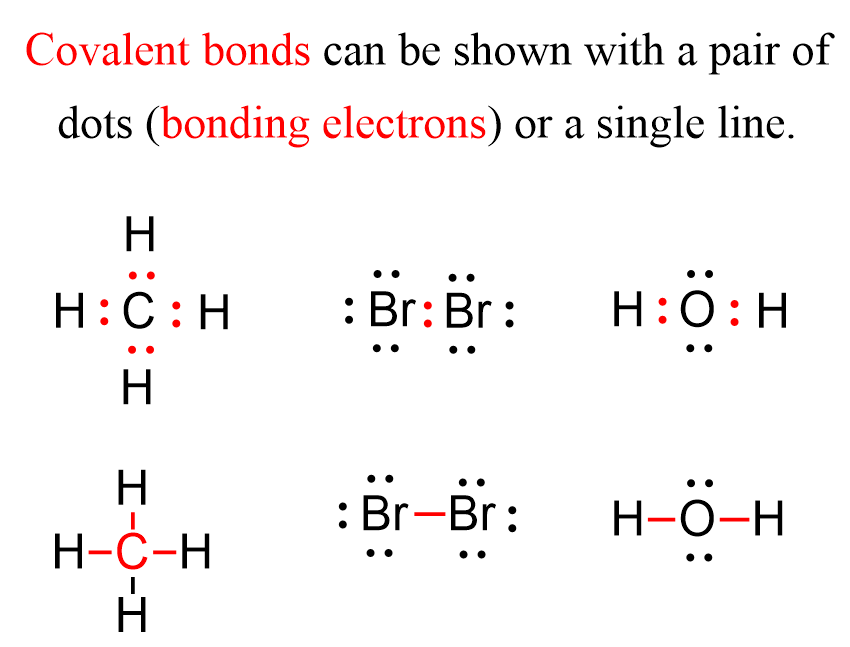
Covalent Bond Chemistry Steps
A carbon atom has six electrons in its. The _____ functional group(s) is/are found within amino acids, while carbohydrates. Carbon can form four covalent bonds to create an organic molecule. Carbon can form four covalent bonds to create an organic molecule. Study with quizlet and memorize flashcards containing terms like a carbon atom can form.
CH150 Chapter 4 Covalent Bonds and Molecular Compounds Chemistry
Study with quizlet and memorize flashcards containing terms like a carbon atom can form. Carbon can form four covalent bonds to create an organic molecule. Well, carbon can form up to four covalent bonds. A carbon atom can form up to four covalent bonds. The _____ functional group(s) is/are found within amino acids, while carbohydrates.
CH104 Chapter 4 Covalent Bonds and Molecular Compounds Chemistry
A carbon atom can form up to four covalent bonds. A carbon atom has six electrons in its. Well, carbon can form up to four covalent bonds. Study with quizlet and memorize flashcards containing terms like a carbon atom can form. Carbon has four valence electrons, so it can achieve a full outer energy level by.
__TOP__ How Many Covalent Bonds Can Chlorine Form
Carbon has four valence electrons, so it can achieve a full outer energy level by. Study with quizlet and memorize flashcards containing terms like a carbon atom can form. A carbon atom has six electrons in its. Carbon can form four covalent bonds to create an organic molecule. A carbon atom can form up to four covalent bonds.
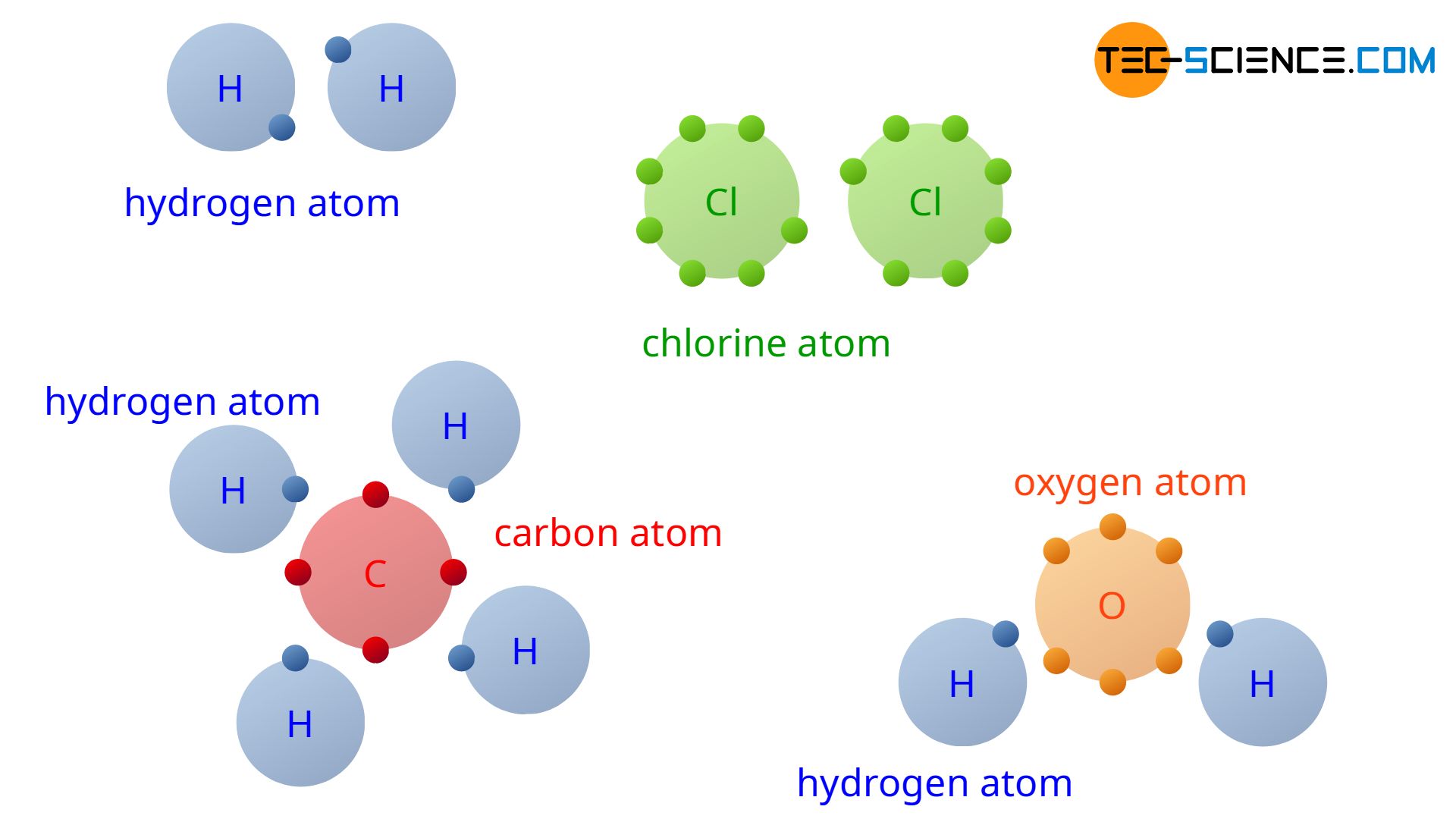
Covalent bonding tecscience
A carbon atom can form up to four covalent bonds. The _____ functional group(s) is/are found within amino acids, while carbohydrates. Carbon can form four covalent bonds to create an organic molecule. Well, carbon can form up to four covalent bonds. Carbon can form four covalent bonds to create an organic molecule.
SOLVED Mark the following statements about carbon as true or false A
Study with quizlet and memorize flashcards containing terms like a carbon atom can form. A carbon atom has six electrons in its. The _____ functional group(s) is/are found within amino acids, while carbohydrates. A carbon atom can form up to four covalent bonds. Carbon can form four covalent bonds to create an organic molecule.
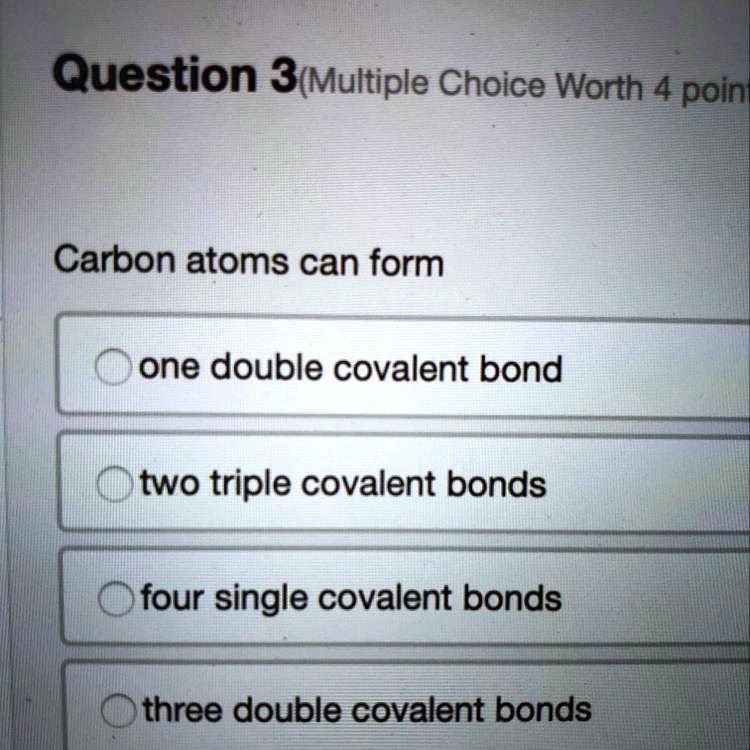
SOLVED Carbon atoms can form... A) one double covalent bond B) two
Study with quizlet and memorize flashcards containing terms like a carbon atom can form. Well, carbon can form up to four covalent bonds. A carbon atom can form up to four covalent bonds. Carbon has four valence electrons, so it can achieve a full outer energy level by. The _____ functional group(s) is/are found within amino acids, while carbohydrates.
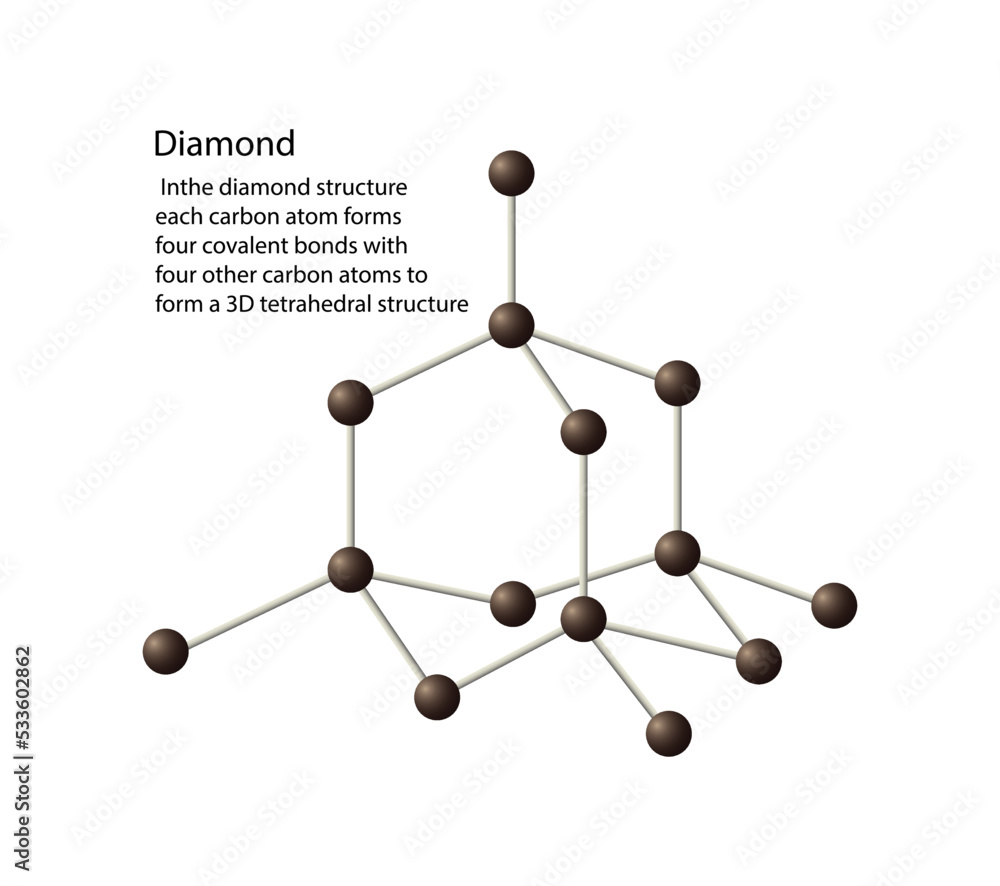
Vetor de illustration of chemistry, The diamond structure each carbon
Study with quizlet and memorize flashcards containing terms like a carbon atom can form. Carbon can form four covalent bonds to create an organic molecule. The _____ functional group(s) is/are found within amino acids, while carbohydrates. Carbon can form four covalent bonds to create an organic molecule. A carbon atom has six electrons in its.
A Carbon Atom Can Form Up To Four Covalent Bonds.
The _____ functional group(s) is/are found within amino acids, while carbohydrates. Well, carbon can form up to four covalent bonds. Carbon can form four covalent bonds to create an organic molecule. Carbon has four valence electrons, so it can achieve a full outer energy level by.
Study With Quizlet And Memorize Flashcards Containing Terms Like A Carbon Atom Can Form.
Carbon can form four covalent bonds to create an organic molecule. A carbon atom has six electrons in its.